SP11 Stress signatures controlling gastrointestinal contractions
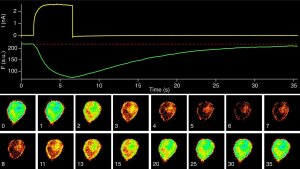
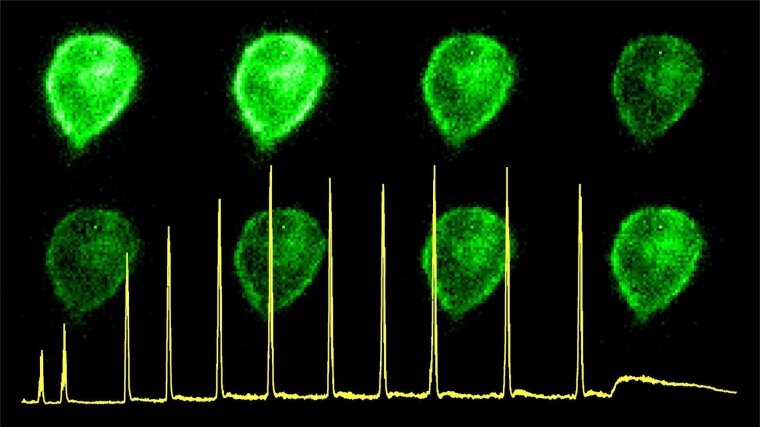
TMEM16A function
Picture: Stefan H. HeinemannProject leader: Stefan H. Heinemann
Doctoral candidate: Rama Hussein
Background and previous work
Malfunctioning of the contractile apparatus of the gut is causative for an array of diseases linked to gut dysmotility. Among them are chronic inflammatory bowel diseases such as Morbus Crohn. Dysbalance of the gastrointestinal microbiome – mostly induced by antibiotic treatment – often worsens the outcome. Although the underlying molecular and cellular mechanisms are clearly multi-facetted, the functional integrity of the contractile systems composed of smooth muscle cells, pacemaking interstitial cells of Cajal, and the myenteric nervous system are required for regular and concerted gastrointestinal contractions. We hypothesize that a set of ion channels, which is required for coordinated contractions, is subject to stress-mediated regulation. In particular this regards Ca2+-dependent Cl– (TMEM16A) and K+ channels (KCNMA1), which are relevant regulators of interstitial cells of Cajal and gut smooth muscle cells, respectively.
Specific aims and working programme
In this PhD project we aim to unravel stress response patterns in the mouse small and large intestine and to associate them to the functioning (or malfunctioning) of the contractile elements, focusing on the ion channels of our expertise. We expect that an adapted activity state of TMEM16A and KCNMA1 is required to maintain healthy contractile rhythms: moderately activated channels being beneficial, while excessive activation may turn into detrimental phenotypes. Besides investigating the impact of stressors on the contractility of gut segments from mice, we will focus on studying the molecular mechanism of select ion channels by stress factors. In the first place these are oxidative events, most likely affecting cysteine and methionine residues. In addition, we evaluate various low-molecular weight compounds released from the gut microbiota. Based on our preliminary experiments, in-depth functional analysis (involving patch-clamp and fluorescence measurements) will be performed with human and murine TMEM16A channels.